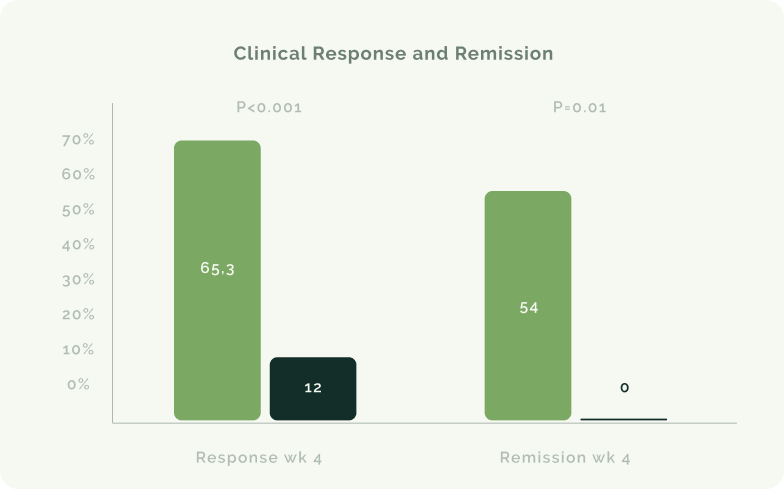
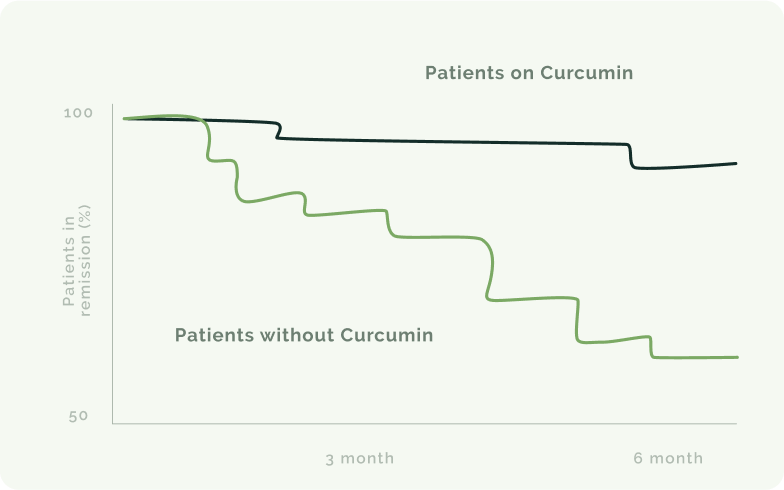
Política de devolución
Dispones de 14 días naturales desde la recepción del producto/s para desistir o devolver el mismo.
No se admite la devolución ni el desistimiento, por razones de protección de la salud e higiene, cuando el envoltorio, embalaje o precinto que protege el producto, haya sido abierto. El producto deberá estar envuelto adecuadamente, con la etiqueta original, no manipulado y con todos sus componentes originales.
Puedes iniciar la devolución del producto contactándonos:
- llamando al teléfono 93 400 67 01
- mediante correo electrónico: info@naturcyte.com.
Enviaremos un mensajero a recoger el producto en la dirección que nos facilites. Entrega el producto al transportista.
El reembolso se realizará por el mismo medio de pago con el que se abonó el producto y por el importe correspondiente. Se realizará antes de que hayan transcurrido 14 días desde la fecha de recepción de la devolución en las oficinas de Adacyte.
Los gastos derivados de la recogida del producto correrán a cargo de Adacyte.
Para cualquier duda, sugerencia, consulta o reclamación sobre la tienda online, puedes dirigirte al Servicio de
atención al cliente.
Atención al cliente:
- Teléfono 93 400 67 01 (en horario de lunes a jueves de 09:00h a 18:00h y viernes de 09:00h a 15:00h)
- Correo electrónico: info@naturcyte.com
- Correo postal en la siguiente dirección:
ADACYTE THERAPEUTICS, S.L.
Calle Jesús Serra Santamans, 5
08174 Sant Cugat del Vallès (Barcelona)
España
RECLAMACIONES
Disponemos de hojas de reclamaciones oficiales a disposición de los consumidores y usuarios, que podrán ser presentadas en la dirección postal y correo electrónico arriba indicados.
Puedes solicitar la hoja de reclamaciones llamando al teléfono de atención al cliente o descargarla directamente aquí: Descargar hojas oficiales.
El Servicio de atención al cliente de Adacyte atenderá y dará respuesta a tu reclamación a la mayor brevedad
posible, y siempre en el plazo máximo de un mes.